最高のコレクション percent yield formula chemistry 305881-Percent yield formula chemistry
The ratio of actual yield to theoretical yield expressed in percentage is called the percentage yield \(\mathrm{percent\ yield = \dfrac{actual\ yield}{theoretical\ yield}\times100}\) Chemical reaction equations give the ideal stoichiometric relationship among reactants and products Thus, the theoretical yield can be calculated fromThe actual yield is the actual amount of product that is produced in a chemical reaction The theoretical yield refers to the amount that should be form when the limiting reagent is completely consumed The actual yield is expressed as a percentage of the theoretical yield This is called the percent yield To find the actual yield, simply multiply the percentage and theoretical yield togetherPrint How to Calculate Percent Yield Definition, Formula & Example Worksheet 1 If the reaction of 125 grams of C6H6O3 reacts in excess of oxygen (O2) and produces 51 grams of H2O, what is the
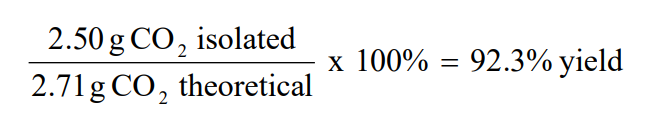
Molecular Formulas And Nomenclature
Percent yield formula chemistry
Percent yield formula chemistry-The percent yield is the ratio of the actual yield to the theoretical yield, expressed as a percentage (1291) Percent Yield = Actual Yield Theoretical Yield × 100 % Percent yield is very important in the manufacture of products Much time and money is spent improving the percent yield for chemical productionYield}\times 100% Percentage yield = \frac {06} {14}\times 100% Percentage yield = 429% The percentage yield of this reaction is 429%, Scientist tries to choose reactions with a high percentage yield or high atom economy


Chemistry Atom Economy And Percentage Yield
So, all you need to put the values into the percent yield formula The percent yieldThe percentage yield is calculated using this equation Percentage yield = \ (\frac {\textup {actual yield}} {\textup {theoretical yield}}\) × 100 The percentage yield can vary from 100% (noThe formula for calculating the percent yield is Percentage yield = mass of actual yield ÷ mass of theoretical yield × 100% Let's assume that you obtained an actual yield of 850 grams Then, the percent yield would be Percentage yield of NaCl = 850 grams ÷ 993 grams × 100% Percentage yield of NaCl = 8559% Since the value of
Learn how to identify the limiting reactant in a chemical reaction and use this information to calculate the theoretical and percent yields for the reaction If you're seeing this message, it means we're having trouble loading external resources on our websiteThe percent yield of this reaction is going to be the actual yield divided by the theoretical yield, multiplied by 100% It's going to be 04 moles over 05 moles times 100% and we have 80% So, the yield of this reaction is 80%WHAT IS THE PERCENT YIELD OF A REACTION?
3 How to determine the percent yield of the reaction considering the limiting reactant Determine the percent yield of the reaction when 770 g of CO 2 are formed from burning 0 moles of C 5 H 12 in 400 moles of O 2 C 5 H 12 8 O 2 → 5 CO 2 6 H 2 OSo, ideally, 336 grams of CaO should have been produced in this reaction This is the theoretical yield However, the problem tells us that only 15 grams were produced 15 grams is the actual yield It is now a simple matter to find percent yieldWhen you look up cheem research papers, the percentage yield of each reaction is written above the arrow The example above shows a threestep method to synthesise Lacosamide, an important medication for epilepsyStep 2 is a stumbling block, with the lowest percentage yield of 37% This means that you only get 37 g of product in the lab, even though you have put in enough reactants to make 100



How To Calculate Percent Yield Definition Formula Example Chemistry Class Video Study Com



Solved The Process Of Using A Balanced Chemical Equation Chegg Com
Percent yield = (actual yield/expected yield) x 100 It can also be written as Percent yield = (actual yield/theoretical yield) x 100Hence, the percent yield is given by Percent Yield = Actual Yield/Theoretical Yield x 100% = 50/7368 x 100% = 6786% The percent yield is 6786%The percent yield of a reaction is the ratio of the actual yield to the theoretical yield CALCULATING % YIELD STEPS 1 Write a balanced chemical equation for the reaction



Percent Yield Calculator 100 Free Calculators Io
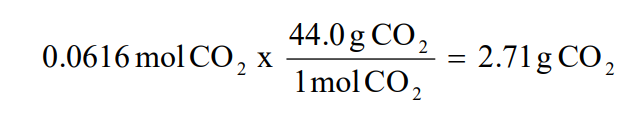


Molecular Formulas And Nomenclature
Theoretical Yield Formula Questions 1 Determine the theoretical yield of H 2 O (in moles) in the following reaction, if 25 moles of hydrogen peroxide are decomposed 2H 2 O 2 → 2H 2 O O 2 Answer In this reaction there is only one reactant (H 2 O 2) so it must be the limiting reactantStoichiometry will be used to determine the moles of water that can be formedHydrazine, N2H4, is an oily liquid used as a rocket fuel It can be prepared in water by oxidizing ammonia with hypochlorite ions 2 NH3g ClOaq > N2H4aq Claq H2Ol When 350 g of ammonia reacted with an excess of hypochlorite ion, 252 g of hydrazine was produced What is the percentage yield of hydrazine?The theoretical yield is a term used in chemistry to describe the maximum amount of product that you expect a chemical reaction could create You need to begin with a balanced chemical equation and define the limiting reactant When you measure the amount of that reactant that you will be using, you can calculate the amount of product



Percent Yield Chemistry Video Clutch Prep
/GettyImages-82845115-1--56a134e33df78cf772686225.jpg)


Stoichiometry Definition In Chemistry
The formula for calculating the percent yield is Percentage yield = mass of actual yield ÷ mass of theoretical yield × 100% Let's assume that you obtained an actual yield of 850 grams Then, the percent yield would be Percentage yield of NaCl = 850 grams ÷ 993 grams × 100% Percentage yield of NaCl = 8559% Since the value ofMixing of exact amounts of reactants such that all are consumed and none left over in a chemical reaction almost never occurs Instead, one of the reactants is usually a limiting reactantSuppose, for example that 100 g of elemental zinc (atomic mass 654) and 80 g of elemental sulfur (atomic mass 3) are mixed and heated undergoing the following reactionPercent yield = actual yield / theoretical yield x 100% percent yield = 15 g / 19 g x 100% percent yield = 79% Usually, you have to calculate the theoretical yield based on the balanced equation In this equation, the reactant and the product have a 11 mole ratio, so if you know the amount of reactant, you know the theoretical yield is the same value in moles (not grams!)


Q Tbn And9gcqsieeu0 Yiv08aomieykfqpxaxi62zyrza1fa2mst1gqjkdvie Usqp Cau



How To Calculate Percent Yield Math Wonderhowto
There are two cases of percent recovery yield below 100% and above 100% The value above 100% is the inaccurate value due to erroneous calculation/weighing The value below 100% is usually the desired valueNext, identify the decimal percentage yield using the chemical formula The actual yield of acetaminophen was reported as 0198g and is then divided by the theoretical yield of 0217g Notice that the actual yield reported was 019 less than the theoretical yield The decimal percentage of percent yield is 0Percent yield represents the ratio between what is experimentally obtained and what is theoretically calculated, multiplied by 100% #"% yield" = ("actual yield")/("theoretical yield") * 100%# So, let's say you want to do an experiment in the lab You want to measure how much water is produced when 1 g of glucose (#C_6H_12O_6#) is burned with enough oxygen
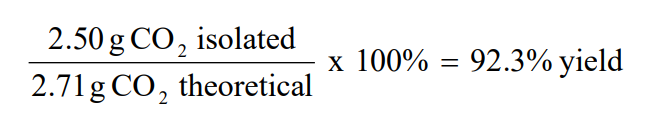


Molecular Formulas And Nomenclature



Limiting Reagent Percent Yield Stoichiometry General Chemistry Lecture 1140 Dr Sundin Uwp
Key Difference – Percent Yield vs Percent Recovery Percent yield is the amount of a compound obtained from a chemical synthesis reaction with respect to the theoretically expected amount This is a percentage value It is used to determine the efficiency of a chemical reaction percent recovery is a term that is often used in organic chemistry regarding recrystallization processesPercent yield = purified percent yield = amount of P (g) theoretical yield (g) •100 Percent yield (if stoichiometry is 11) = amount of P (mol) amount of LR (mol) •100 Complicated balanced equations are uncommon in organic chemistry Many organic reactions have a stoichiometry of 11 in the balanced equationPercent yield is a measure of how well the reaction proceeded to completion The formula for percent yield is the experimental yield divided by the calculated (theoretical yield) 4



Solved For The Balansed Chemical Equations Below Find Th Chegg Com



Calculating Reaction Yield And Percentage Yield From A Limiting Reactant Science Class Video Study Com
How do we find the percentage here?Worked Example of Percentage Yield Calculations Calculating Mass of Product from Yield Question Ammonia can be produced from hydrogen gas and nitrogen gas according to the equation below N 2(g) 3H 2(g) ⇋ 2NH 3(g) Calculate the mass of ammonia produced if 168 g of nitrogen gas produces a yield of 45%6O 2 C 6 H 12 O 6 6 → CO 2 6H 2 O After balancing the equation, now we know that after the actual reaction if there is no loss of reactants, our product will be six molecules of carbon dioxide and six molecules of water It is the theoretical yield However, it is impossible for a ration to give 100 percent yield



How To Calculate Percent Yield Science Chemistry Chemistry Class Chemistry


2
Percent yield = purified percent yield = amount of P (g) theoretical yield (g) •100 Percent yield (if stoichiometry is 11) = amount of P (mol) amount of LR (mol) •100 Complicated balanced equations are uncommon in organic chemistry Many organic reactions have a stoichiometry of 11 in the balanced equationThe percent yield be the actual yield (392) divided by the theoretical yield (i calculated 450) 392/450 equals 87 multiply by 100 and the percent yield is 87% What relationship s used toChemistry Journal 506 Percent Yield Driving Question How do chemists use percentages and stoichiometry to understand the amount of products produced during chemical reactions?



Magnesium Oxide Percent Yield Lab Report Schoolworkhelper



A Level Chemistry Ocr Salters Yield Wikibooks Open Books For An Open World
Next, identify the decimal percentage yield using the chemical formula The actual yield of acetaminophen was reported as 0198g and is then divided by the theoretical yield of 0217g Notice that the actual yield reported was 019 less than the theoretical yield The decimal percentage of percent yield is 0To use this formula for percent yield, you need to make sure that your actual yield and theoretical yield are in the same units If the actual yield is in grams, then theoretical yield also needsPercentage yield = (15 ÷ ) × 100 = 75% Losing product A 100 per cent yield means that no product has been lost, while a 0 per cent yield means that no product has been made
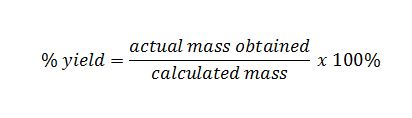


Moles Percentage Purity And Yield John Vagabond S Physics And Chemistry Blog
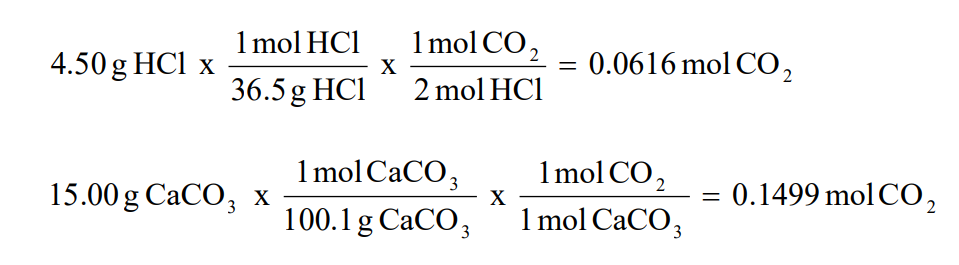


Molecular Formulas And Nomenclature
Organic Chemistry FriedelCrafts Alkylation of Dimethoxybenzene Description & Background 0027 mol salicylic acid X g = 372g mol 3 Divide the number of grams of product obtained experimentally, by the number of grams obtained in the theoretical yield calculations and multiply by 100 to calculate the percent yieldExample Consider a 352g sample of CaCO 3 (9987% pure) in a flask and a 1000 mL sample of vinegar (5% acidity) in a graduated cylinderBut the question states that the actual yield is only 3791 g of sodium sulfate With these two pieces of information, you can calculate the percent yield using the percentyield formula So, you find that 8137% is the percent yield



How To Calculate Percent Yield Definition Formula Example Chemistry Class Video Study Com



Calculating Actual Yield Given The Percent Yield Youtube
Organic Chemistry FriedelCrafts Alkylation of Dimethoxybenzene Description & Background 0027 mol salicylic acid X g = 372g mol 3 Divide the number of grams of product obtained experimentally, by the number of grams obtained in the theoretical yield calculations and multiply by 100 to calculate the percent yieldThe extent to which a reaction's theoretical yield is achieved is commonly expressed as its percent yield percent yield = actual yield theoretical yield ×100% percent yield = actual yield theoretical yield × 100 %Percent yield or percentage yield is the ratio of the actual yield and the theoretical yield of a chemical reaction The experimental yield is divided by the theoretical yield and multiplied by 100 to be calculated as the percent yield If the theoretical yield and the experimental yield are same then the percent yield will be 100%



Chemistry 12 3 Limiting Reagent And Percent Yield Ppt Download



Howto How To Find Percentage Yield In Chemistry
So, the percent yield = 1212 ÷ 1305 × 100% = 929% How to calculate the percent yield of a chemical reaction?The percentage yield can vary from 100% (no product has been lost) to 0% (no product has been made) Worked example Copper oxide reacts with sulfuric acid to make copper sulfate and waterPercentage yield = \frac {Actual\;



Percent Yield Formula Chem Service Unavailable In Eu Region



How To Calculate Percent Yield 3 Ways To Solve Chemistry Problems Tripboba Com
Hydrazine, N2H4, is an oily liquid used as a rocket fuel It can be prepared in water by oxidizing ammonia with hypochlorite ions 2 NH3g ClOaq > N2H4aq Claq H2Ol When 350 g of ammonia reacted with an excess of hypochlorite ion, 252 g of hydrazine was produced What is the percentage yield of hydrazine?Percent Yield The amount of product that may be produced by a reaction under specified conditions, as calculated per the stoichiometry of an appropriate balanced chemical equation, is called the theoretical yield of the reaction In practice, the amount of product obtained is called the actual yield, and it is often less than the theoretical yield for a number of reasonsThis video shows you how to calculate the theoretical and percent yield in chemistry The theoretical yield is the maximum amount of product that can be pro



Calculating Percent Recovery Percent Yield



Percentage Yield By David Gabb
To express the efficiency of a reaction, you can calculate the percent yield using this formula %yield = (actual yield/theoretical yield) x 100 A percent yield of 90% means the reaction was 90% efficient, and 10% of the materials were wasted (they failed to react, or their products were not captured) Steps



Yield Calculations Faculty Staff Sites



Quantitative Chemistry Secondary Science 4 All



Organic Chemistry Lab I Chem 237 Experiment 2 Recrystallization Flashcards Quizlet



Magnesium Oxide Percent Yield Lab Report Schoolworkhelper



Chemistry 101 Calculating Reactant Given Percent Yield And Actual Product Youtube



Percent Yield Calculator



Reaction Percent Yield Introduction And Practice Exercises



Limiting Reactant And Reaction Yields Article Khan Academy



Percentage Yield Lab Answers Schoolworkhelper



Reaction Percent Yield Introduction And Practice Exercises



Calculating Reaction Yield And Percentage Yield From A Limiting Reactant Science Class Video Study Com



Howto How To Find Percent Yield Without Actual Yield


Q Tbn And9gcrynatxciaobbvt Edbzcqmlgnhpv Acw5cqav2pwzm1fudi Ih Usqp Cau



How To Calculate Percent Yield Definition Formula Example Chemistry Class Video Study Com



Percent Yield And Percent Error Calculations Science Chemistry Percent Yield Percent Error Showme



How To Calculate Theoretical Yield 12 Steps With Pictures



Stoichiometry Solving Percent Yield Stoichiometry Problems Youtube



General Chemistry I Calculation Of The Formula Of Chegg Com



Question Video Calculating The Percentage Yield Of The Recreation Of Aqueous Copper Sulfate With Zinc Metal Nagwa


Calculating Percentage Yield


Www Assignmentexpert Com Homework Answers Chemistry Answer Pdf


Limiting Reagents



Solution Magnesium Oxide Can Be Made By H Clutch Prep


Chemistry Atom Economy And Percentage Yield



How To S Wiki How To Calculate Percentage Yield


Atom Economy Yield



Calculating Percent Yield Hey Chemistry


The Stoichiometry Of Product Formation And Percent Yield



Yields Introductory Chemistry



Percent Yield Percent Purity Solutions Examples Videos



How To Calculate Percentage Yield In Chemistry How To Wiki



Solved Determine The Limiting Reactant Express Answer As Chegg Com



How To Calculate Theoretical Yield And Percent Yield Youtube



A Level Chemistry Ocr Salters Yield Wikibooks Open Books For An Open World


Williamwithin Com Uploads 1070 Theoretical Yiel



How To Calculate The Percent Yield And Theoretical Yield Youtube


3
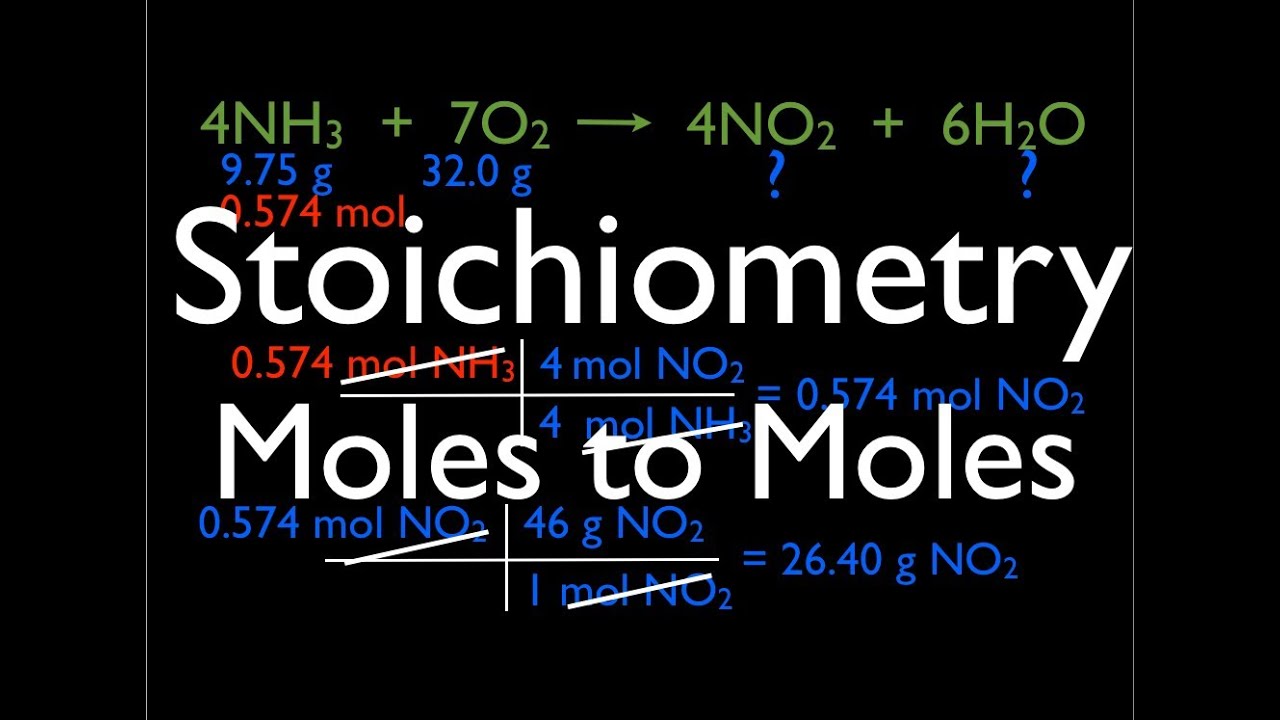


Reaction Stoichiometry Boundless Chemistry
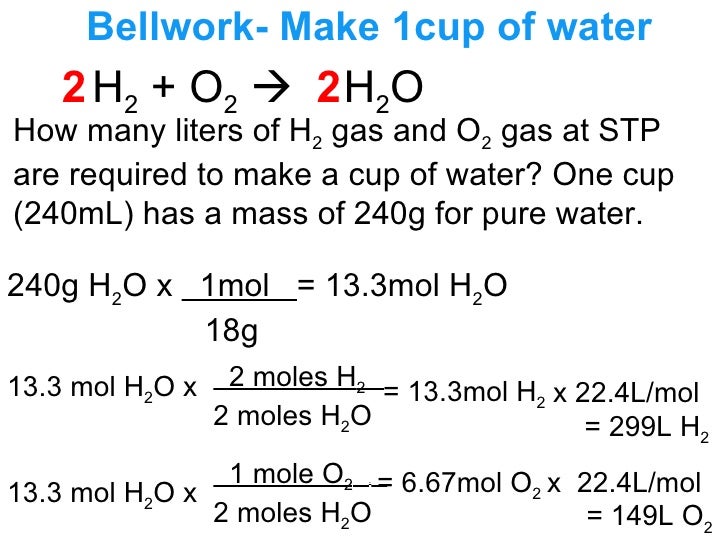


Lecture 12 3 Limiting Reagents And Percent Yield


Swhschemistry Weebly Com Uploads 7 8 3 2 Lt 7 2 Notes Pdf



Percent Yield Calculations Percent Yield Calculations 1 Balance This Equation And State Which Of The Six Types Of Reaction Is Taking Place Mg Hno3 Course Hero



Percent Yield Concept Chemistry Video By Brightstorm
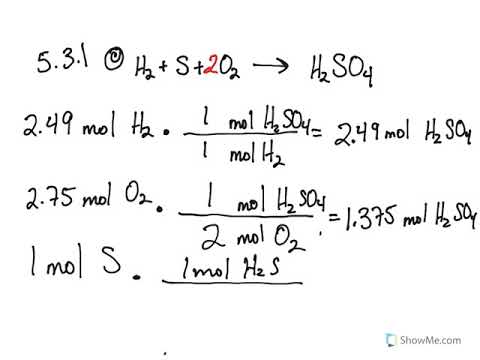


5 3 Calculating Reaction Yields Problems Chemistry Libretexts



Mole Stoich Resources



Pin By Jesus Gomez On Estequiometria Chemistry Class Science Chemistry Worksheets



Yield Calculations Faculty Staff Sites



How To Calculate Percent Yield In Chemistry 15 Steps



Calculating Percent Yield Hey Chemistry
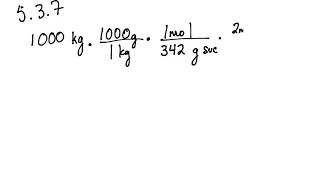


5 3 Calculating Reaction Yields Problems Chemistry Libretexts



Chemistry Calculations Percent Yield And Atom Economy



Percent Yield Made Easy Stoichiometry Tutorial Part 4 Youtube



10 Atom Economy Sjp1618gcsechem



Stoichiometry And Percent Yield Examples Solutions Worksheets Videos Games Activities



How To Calculate Percent Yield Definition Formula Example Chemistry Class Video Study Com



Theoretical Actual And Percent Yield Problems Chemistry Tutorial Youtube



Percent Yield And Percent Error Calculations Science Chemistry Percent Yield Percent Error Showme



Percentage Yield Lab Answers Schoolworkhelper



Percent Yield Chemistry Video Clutch Prep
:max_bytes(150000):strip_icc()/148302528-56a12f323df78cf77268383a.jpg)


Percent Yield Definition And Formula
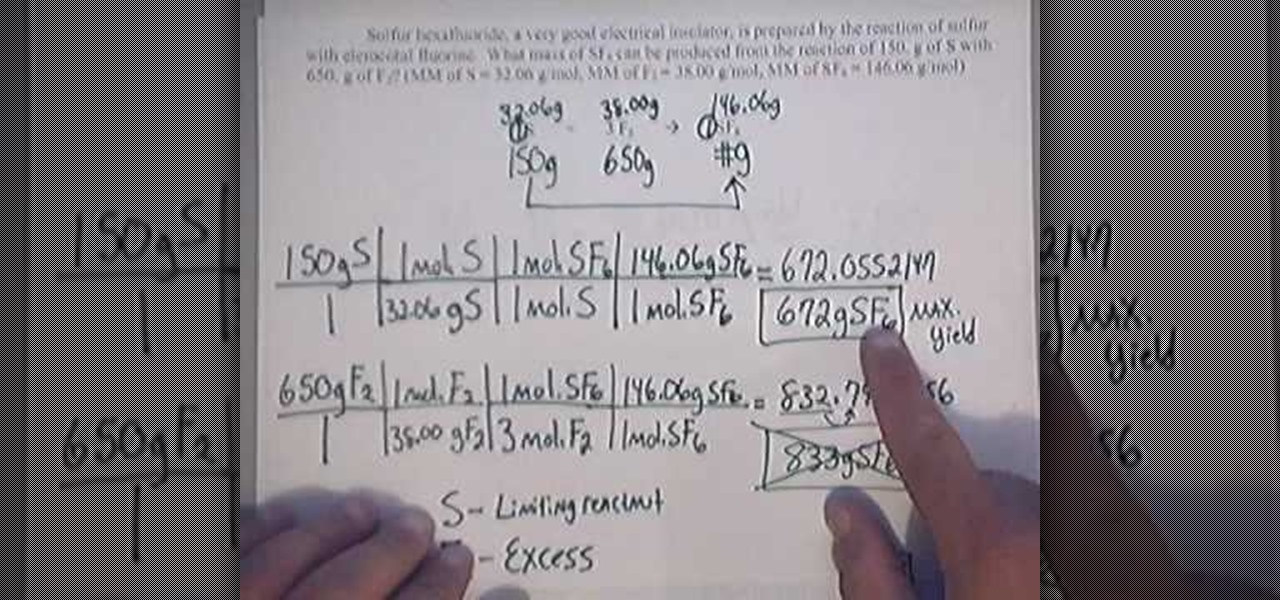


How To Calculate Percent Yield Math Wonderhowto



Reaction Percent Yield Introduction And Practice Exercises


Q Tbn And9gcrjyqnm Hsuw1zecxl5b8r5elmfwvpiet 7gqpwpfgze5a6h7ox Usqp Cau



Percent Yield Calculation Page 1 Line 17qq Com


Calculation Of Theoretical Yield Organic Chemistry I 212 01


Calculating Percentage Yield



Calculating Percent Yield Hey Chemistry



I Redd It Hy37q3l91sp41 Jpg



Yields Introductory Chemistry



18 Percentage Yield
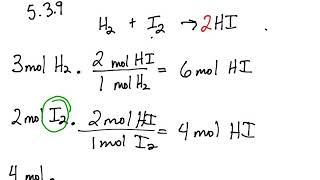


5 3 Calculating Reaction Yields Problems Chemistry Libretexts



Percent Yield Tutorial Explained Practice Problems Crash Chemistry Academy Youtube


Chemistry Atom Economy And Percentage Yield



Quantitative Chemistry Theoretical And Percent Yield


Atom Economy Yield



Percent Yield Calculator



Stoichiometry And Percent Yield Examples Solutions Worksheets Videos Games Activities



What Would Be My Percent Yield If I Used 1 6 G 2 Methyl 2 Butanol To Create A Mixture Homeworklib
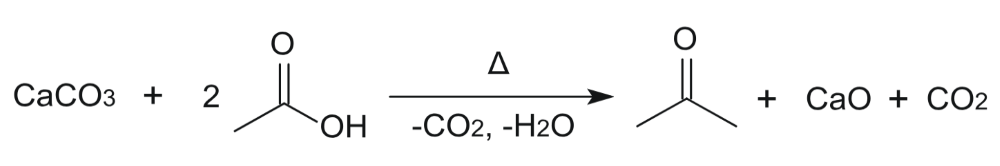


Percent Yield Calculator


コメント
コメントを投稿